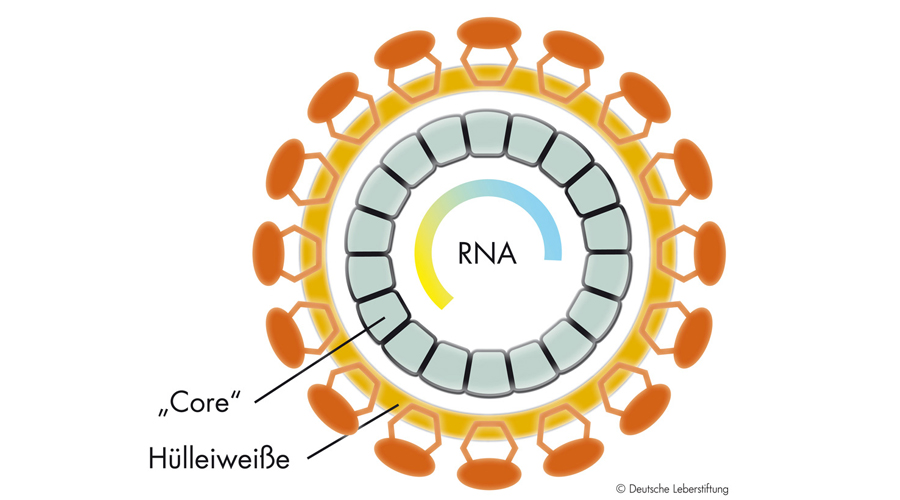
Publikationen 2025
Transient interferon-driven NK cell activation in acute hepatitis C B. Strunz, Q. Zhan, T. Khera, J. Hengst, M. Jankovic, K. Deterding, et al. J Infect Dis 2025 Accession Number: 41453396 DOI: 10.1093/infdis/jiaf654
Impact of distinct antiviral treatment regimens on the long-term outcome after HCV cure – Data from the German Hepatitis C-Registry (DHC-R) V. Ohlendorf, Y. Serfert, P. Buggisch, S. Mauss, H. Klinker, G. Teuber, et al. Z Gastroenterol 2025 Vol. 63 Issue 5 Pages 486-496 Accession Number: 40360141 DOI: 10.1055/a-2543-5205
Distinct Inflammatory Imprint in Non-Cirrhotic and Cirrhotic Patients Before and After Direct-Acting Antiviral Therapy M. Witte, C. Oltmanns, J. Tauwaldt, H. Schmaus, J. Mischke, G. Grabert, et al. Clin Mol Hepatol 2025 Accession Number: 40462644 DOI: 10.3350/cmh.2025.0292
Publikationen 2024
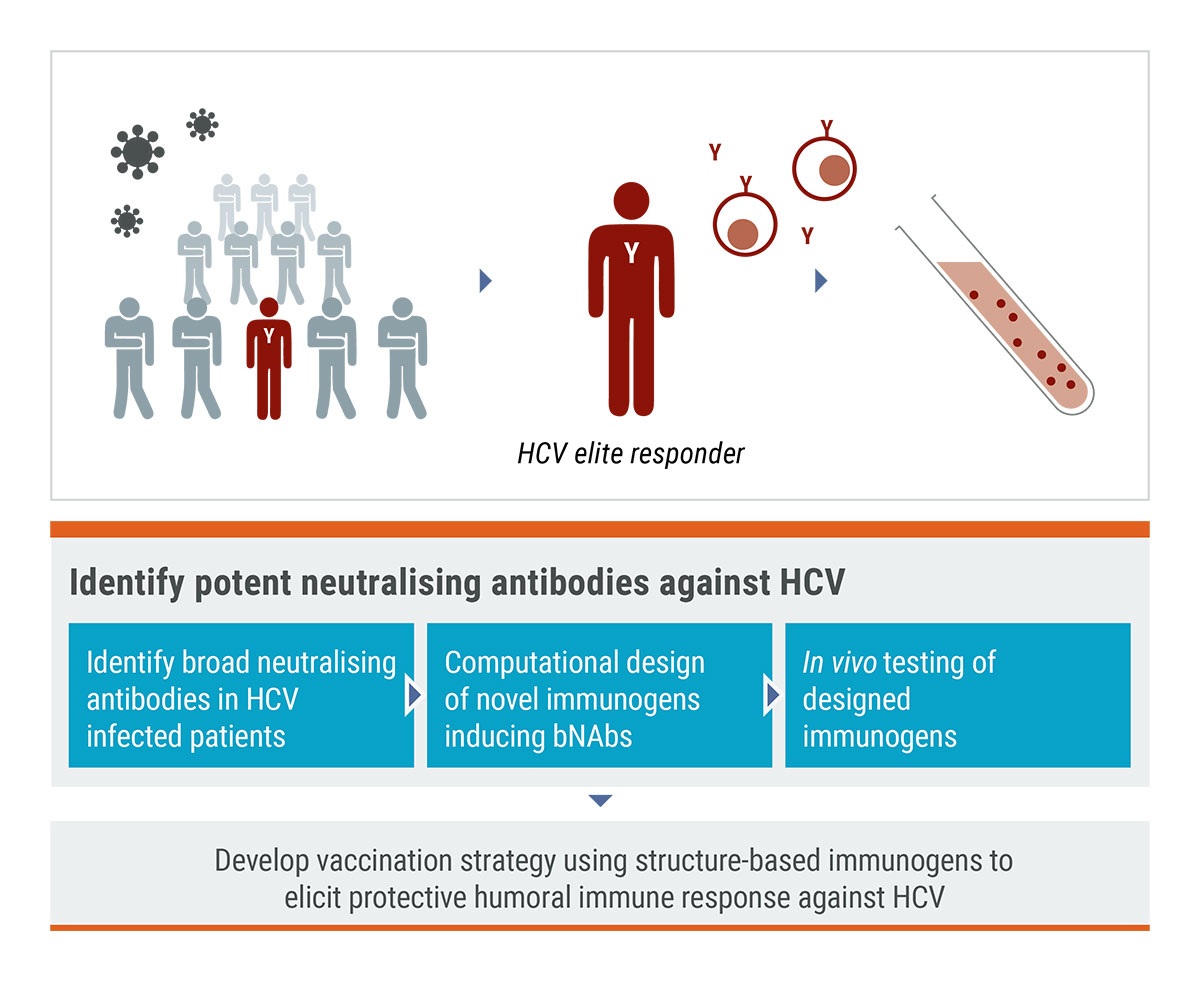
Epitope-focused immunogens targeting the hepatitis C virus glycoproteins induce broadly neutralizing antibodies K. Nagarathinam, A. Scheck, M. Labuhn, L. J. Ströh, E. Herold, B. Veselkova, et al. Sci Adv 2024 Vol. 10 Issue 49 Pages eado2600 Accession Number: 39642219 PMCID: PMC11623273 DOI: 10.1126/sciadv.ado2600
Impending HCC diagnosis in patients with cirrhosis after HCV cure features a natural killer cell signature S. A. Engelskircher, P. C. Chen, B. Strunz, C. Oltmanns, T. Ristic, S. Owusu Sekyere, et al. Hepatology 2024 Vol. 80 Issue 1 Pages 202-222 Accession Number: 38381525 PMCID: PMC11191062 DOI: 10.1097/hep.0000000000000804
Unraveling the dynamics of hepatitis C virus adaptive mutations and their impact on antiviral responses in primary human hepatocytes. Frericks N, Brown RJP, Reinecke BM, Herrmann M, Brüggemann Y, Todt D, Miskey C, Vondran FWR, Steinmann E, Pietschmann T, Sheldon J. J Virol. 2024 Feb 6:e0192123. doi: 10.1128/jvi.01921-23. Online ahead of print. PMID: 38319104
Elevation of S2-bound α1-acid glycoprotein is associated with chronic hepatitis C virus infection and hepatocellular carcinoma. Oltmanns, C., B. Bremer, L. Kusche, P. Stål, R. Zenlander, J. Tauwaldt, I. Rydén, P. Påhlsson, M. Cornberg, and H. Wedemeyer. 2024. J Viral Hepat.
Publikationen 2023
Proteomics reveals a global phenotypic shift of NK cells in HCV patients treated with direct-acting antivirals. Bi W, Kraft A, Engelskircher S, Mischke J, Witte M, Klawonn F, van Ham M, Cornberg M, Wedemeyer H, Hengst J, Jänsch L. Eur J Immunol. 2023 Jul 29:e2250291.
Structural insights into hepatitis C virus neutralization. Ströh LJ, Krey T. Curr Opin Virol. 2023 Mar 29;60:101316.
Immunological scars after cure of hepatitis C virus infection: Long-HepC? Cornberg M, Mischke J, Kraft AR, Wedemeyer H. Curr Opin Immunol. 2023 Apr 10;82:102324.
Controlled Attenuation Parameter Is Associated with a Distinct Systemic Inflammatory Milieu after Clearance of HCV Infection. Du Y, Khera T, Liu Z, Tudrujek-Zdunek M, Dworzanska A, Cornberg M, Xu CJ, Tomasiewicz K, Wedemeyer H. Biomedicines. 2023 May 25;11(6):1529. doi: 10.3390/biomedicines11061529. PMID: 37371624; PMCID: PMC10295384.
Publikationen 2022
Development approaches for vaccines against hepatitis C virus infections. Bankwitz D, Krey T, Pietschmann T. Bundesgesundheitsblatt Gesundheitsforschung Gesundheitsschutz. 2022 Feb;65(2):183-191. German. doi: 10.1007/s00103-021-03477-9. Epub 2022 Jan 11. PMID: 35015104; PMCID: PMC8749110.
Analysis of antibodies from HCV elite neutralizers identifies genetic determinants of broad neutralization. Weber T, Potthoff J, Bizu S, Labuhn M, Dold L, Schoofs T, Horning M, Ercanoglu MS, Kreer C, Gieselmann L, Vanshylla K, Langhans B, Janicki H, Ströh LJ, Knops E, Nierhoff D, Spengler U, Kaiser R, Bjorkman PJ, Krey T, Bankwitz D, Pfeifer N, Pietschmann T, Flyak AI, Klein F. Immunity.
The Human Liver-Expressed Lectin CD302 Restricts Hepatitis C Virus Infection. Reinecke B, Frericks N, Lauber C, Dinkelborg K, Matthaei A, Vondran FWR, Behrendt P, Haid S, Brown RJP, Pietschmann T. J Virol.
A Hepatitis C virus genotype 1b post-transplant isolate with high replication efficiency in cell culture and its adaptation to infectious virus production in vitro and in vivo. Heuss C, Rothhaar P, Burm R, Lee JY, Ralfs P, Haselmann U, Ströh LJ, Colasanti O, Tran CS, Schäfer N, Schnitzler P, Merle U, Bartenschlager R, Patel AH, Graw F, Krey T, Laketa V, Meuleman P, Lohmann V. PLoS Pathog.
An Equine Model for Vaccination against a Hepacivirus: Insights into Host Responses to E2 Recombinant Protein Vaccination and Subsequent Equine Hepacivirus Inoculation M. Badenhorst, A. Saalmüller, J. M. Daly, R. Ertl, M. Stadler, C. Puff, et al. Viruses 2022 Vol. 14 Issue 7 Pages 1401 DOI: 10.3390/v14071401
Metamizole-Associated Risks in Decompensated Hepatic Cirrhosis B. Schulte, T. L. Tergast, M. Griemsmann, D. Menti, N. Deveci, J. Kahlhöfer, et al. Dtsch Arztebl Int 2022 Vol. 119 Issue 41 Pages 687-693 Accession Number: 35912424 PMCID: PMC9830680 DOI: 10.3238/arztebl.m2022.0280
Publikationen 2021
Initial HCV infection of adult hepatocytes triggers a temporally structured transcriptional program containing diverse pro- and anti-viral elements B. Tegtmeyer, G. Vieyres, D. Todt, C. Lauber, C. Ginkel, M. Engelmann, et al. Journal of Virology 2021 DOI: 10.1128/JVI.00245-21
EpitopeVec: Linear Epitope Prediction Using Deep Protein Sequence Embeddings. Bioinformatics.Bahai A, Asgari E, Mofrad MRK, Kloetgen A, McHardy AC.
Hepatitis C reference viruses highlight potent antibody responses and diverse viral functional interactions with neutralising antibodies. Bankwitz D, Bahai A, Labuhn M, Doepke M, Ginkel C, Khera T, Todt D, Ströh LJ, Dold L, Klein F, Klawonn F, Krey T, Behrendt P, Cornberg M, McHardy AC, Pietschmann T. Gut. 2020
Publikationen 2020
Liver-expressed Cd302 and Cr1l limit hepatitis C virus cross-species transmission to mice. Brown RJP, Tegtmeyer B, Sheldon J, Khera T, Anggakusuma, Todt D, Vieyres G, Weller R, Joecks S, Zhang Y, Sake S, Bankwitz D, Welsch K, Ginkel C, Engelmann M, Gerold G, Steinmann E, Yuan Q, Ott M, Vondran FWR, Krey T, Ströh LJ, Miskey C, Ivics Z, Herder V, Baumgärtner W, Lauber C, Seifert M, Tarr AW, McClure CP, Randall G, Baktash Y, Ploss A, Thi VLD, Michailidis E, Saeed M, Verhoye L, Meuleman P, Goedecke N, Wirth D, Rice CM, Pietschmann Sci Adv.
HCV Glycoprotein Structure and Implications for B-Cell Vaccine Development. Ströh LJ, Krey T. Int J Mol Sci.
Global and local envelope protein dynamics of hepatitis C virus determine broad antibody sensitivity. Augestad EH, Castelli M, Clementi N, Ströh LJ, Krey T, Burioni R, Mancini N, Bukh J, Prentoe J. Sci Adv.
De novo protein design enables the precise induction of RSV-neutralizing antibodies. Sesterhenn F, Yang C, Bonet J, Cramer JT, Wen X, Wang Y, Chiang CI, Abriata LA, Kucharska I, Castoro G, Vollers SS, Galloux M, Dheilly E, Rosset S, Corthésy P, Georgeon S, Villard M, Richard CA, Descamps D, Delgado T, Oricchio E, Rameix-Welti MA, Más V, Ervin S, Eléouët JF, Riffault S, Bates JT, Julien JP, Li Y, Jardetzky T, Krey T, Correia BE. Science.
Efficient acute and chronic infection of stem cell-derived hepatocytes by hepatitis C virus. Carpentier A, Sheldon J, Vondran FWR, Brown RJ, Pietschmann T. Gut.